Cardiovascular System
The cardiovascular system is also called the circulatory system or the blood-vascular system. It is primarily responsible for transporting oxygen among many other important substances and nutrients throughout the body. The heart, a powerful pump, utilizes an intricate and extensive network of blood vessels to perform this function.
Anatomy of the Heart


Anatomical Location of the Heart
- Snugly enclosed within the middle mediastinum (medial cavity of thorax), which contains the
- Heart
- Pericardium
- Great Vessels
- Trachea
- Esophagus
- Middle Mediastinum – located in the inferior mediastinum (lower than the sternal angle)
- Extends obliquely from 2nd rib → 5th intercostal space
- Anterior to Vertebrae
- Posterior to Sternum
- Flanked by 2 lungs
- Rests on the diaphragm
- 2/3 of its mass lies to the LHS of the midsternal line
The Pericardium (Coverings of the Heart)
- A double-walled sac
- Contains a film of lubricating serous fluid
- 2 Layers of Pericardium
- Fibrous Pericardium
- Tough, dense connective tissue
- Protects the heart
- Anchors it to surrounding structures
- Prevents overfilling of the heart – if fluid builds up in the pericardial cavity, it can inhibit effective pumping. (Cardiac Tamponade)
- Serous Pericardium (one continuous sheet with ‘2 layers’)
- Parietal Layer – Lines the internal surface of the fibrous pericardium
- Visceral Layer – (aka Epicardium) Lines the external heart surface
- Fibrous Pericardium
Layers of the Heart Wall
- Epicardium
- Visceral layer of serous pericardium
- Myocardium
- Muscle of the heart
- The layer that ‘contracts’
- Endocardium
- Lines the chambers of the heart (endothelial cells)
- Prevents clotting of blood within the heart
- Forms a barrier between the O2 hungry myocardium and the blood (blood is supplied via the coronary system)
Fibrous Skeleton of the Heart
- The network of connective tissue fibers (collagen & elastin) within the myocardium
- Anchors the cardiac muscle fibers + valves + great vessels.
- Reinforces the myocardium
- Provides electrical isolation
- 2 parts
- Septum
- Flat sheets separating atriums, ventricles & left and right sides of the heart
- Electrically isolates the left & right sides of the heart (connective tissue = non-conductive)
- Important for cardiac cycle
- Interatrial septum/atrioventricular septum/interventricular septum
- Ring
- Rings around great vessel entrances & valves
- Stops stretching under pressure
- Septum
Chambers & Associated Great Vessels
2 Atria (Superior)
- Atrium = entryway
- On the superior aspect of the heart (above the ventricles)
- Each has a small, protruding appendage called auricles – increasing atrial volume
- Separated by Atrial Septum (Site of Fetal Shunt Foramen Ovale)
- Right Atrium
- Ridged internal anterior wall – due to muscle bundles called pectinate muscles
- Blood enters via 3 veins
- Superior Vena Cava
- Inferior Vena Cava
- Coronary Sinus (collects blood draining from the myocardium)
- Left Atrium
- Blood enters via
- The 4 pulmonary veins (O2 blood)
[rml_read_more]
2 Ventricles (Inferior)
- Vent = underside
- Thick, muscular Discharging Chambers
- The ‘pumps’ of the heart
- Trabeculae Carneae (crossbars of flesh) line the internal walls
- Papillary Muscles play a role in valve function
- Right Ventricle
- Most of heart’s Anterior Surface
- Thinner – responsible for the Pulmonary Circulation – Via Pulmonary Trunk
- Left Ventricle
- Most of the heart’s Postero-Inferior Surface
- Thicker – it is responsible for the Systemic Circulation – Via Aorta
Landmarks of the Heart
- Coronary Sulcus (Atrioventricular Groove)
- Encircles the junction between the Atria & Ventricles like a ‘Crown’ (Corona)
- Cradles the Coronary Arteries (R&L), Coronary Sinus, & Great Cardiac Vein
- Anterior Interventricular Sulcus
- Cradles the Anterior Interventricular Artery (Left Anterior Descending Artery)
- Separates the right & left Ventricles anteriorly
- Continues as the posterior Interventricular Sulcus
- Posterior Interventricular Sulcus
- Cradles the Posterior Descending Artery
- Continuation of the Anterior Interventricular Sulcus
- Separates the right & left ventricles posteriorly
Pathway of Blood through the Heart
The right side
- pumps blood through the pulmonary circuit (to the lungs and back to the left side of the heart)
- blood flowing through the pulmonary circuit gains oxygen and loses carbon dioxide, indicated by the color change from blue to red
The left side
- pumps blood via the systemic circuit to all body tissues and back to the right side of the heart
- blood flowing through the systemic circuit loses oxygen and picks up carbon dioxide (red to blue color change)
Coronary Circulation
- The myocardium’s own blood supply
- The shortest circulation in the body
- Arteries lie in epicardium – prevents the contractions inhibiting blood flow
- There is a lot of variation among different people
Arterial Supply
- Encircle the heart in the coronary sulcus
- Aorta → Left & Right coronary arteries
- Left Coronary Artery → 2 Branches:
- Anterior Interventricular Artery (aka. Left Anterior Descending Artery or LAD
- Follows the Anterior InterVentricular Sulcus
- Supplies Apex, Anterior LV, Anterior 2/3 of IV-Septum
- Circumflex Artery
- Follows the Coronary Sulcus (aka. AtrioVentricular Groove)
- Supplies the Left Atrium + Lateral LV
- Right Coronary Artery → 2 (‘T-junction) Branches:
- Marginal Artery:
- Serves the Myocardium Lateral RHS of Heart
- Marginal Artery:
- Posterior Interventricular Artery:
- Supplies posterior ventricular walls
- Anastomoses with the Anterior Interventricular Artery (LAD)
Venous Drainage
- Venous blood – collected by the Cardiac Veins (empties into the right atrium)
- Great Cardiac Vein (in Anterior InterVentricular Sulcus)
- Middle Cardiac Vein (in Posterior InterVentricular Sulcus)
- Small Cardiac Vein (along Right inferior Margin)
Heart Valves
Ensure unidirectional flow of blood through the heart
2x Atrioventricular (AV) (Cuspid) Valves
- Location → at the 2 atrial-ventricular junctions
- Function → prevent backflow into the atria during contraction of ventricles
- Chordae tendineae (tendinous cords) “heart strings” – attached to each valve flap
- Anchor the cusps to the Papillary Muscles protruding from ventricular walls.
- Papillary muscles contract before the ventricle to tension the chordae tendineae
- Prevent inversion of valves under ventricular contraction
- Anchor the cusps to the Papillary Muscles protruding from ventricular walls.
- Tricuspid valve (right)
- 3 flexible ‘cusps’ (flaps of endocardium + Conn. Tissue)
- Mitral valve (left)
- (resembles the 2-sided bishop’s mitre [hat])
2x Semilunar (SL) Valves
- Located at the bases of both large arteries issuing from the ventricles
- Each consists of 3 pocket-like cusps resembling a crescent moon (semilunar = half moon)
- Open under Ventricular Pressure
- Pulmonary Valve
- Between Right Ventricle & Pulmonary Trunk
- Aortic Valve
- Between Left Ventricle & Aorta


Valve Positions during ventricular contraction (left) and relaxation (right)
Valve Sounds
- “Lubb”
- Sound of AV valve closure
- M1 = Mitral component
- T1 = Tricuspid component
- “Dupp”
- Sound of SL valve closure
- A2 = Aortic component
- P2 = Pulmonary component
Electrophysiology of the Heart
The Heartbeat
- Heart is a muscle and requires
- O2,
- Nutrients, and
- Action Potentials to function
- However, these neural signals don’t come from the brain;
- Rather, the heart has its own conduction systems
- allow heart to contract autonomously
- Hence why a transplanted heart still operates (if provided with O2 and nutrients)
- Cardiac Activity is coordinated:
- To be effective, the atria & ventricles must contract in a coordinated manner
- This activity is coordinated by the heart’s Conduction Systems
- The entire heart is electrically connected by:
- Gap junctions
- Allow action potentials to move from cell to cell
- Intercalated discs
- Support synchronized contraction of cardiac tissue
- Gap junctions
The Heart’s Conduction Systems
SA Node → AV Node → Bundle of His → R&L Bundle Branches → Purkinje Fibers → Myocyte Contraction
Conductile Cardiac Cell Physiology (SA/AV Node Cells)
- Action Potentials: Slow ‘Pacemaker’ Type
- Have UNSTABLE Resting Membrane Potentials → spontaneous electrical activity
- Spontaneously depolarize to threshold
- This gradual depolarisation is called a ‘Prepotential’
- Due to leaky Na+ membrane ion channels
- Therefore – firing frequency depends on Na+ movement
- Depolarisation:
- Once threshold is reached, Ca2+ channels open
- → Influx of Ca+
- → Causes an action potential
- Repolarisation:
- Once peak MP is reached, Ca+ channels close, K+ channels open
- → K+ Efflux makes MP more negative
- Spontaneously depolarize to threshold
- → Causes repolarisation
- (Na+ brings to threshold, but Ca+ is responsible for Depolarisation.)
- With a Hierarchy of control over the heart
- Hierarchy based on natural intrinsic rate (fastest node (SA node) takes control)
Contractile Cardiac Cell Physiology (Purkinje Fibers & Myocytes)
- Action Potentials: Fast ‘Non-Pacemaker’ Type
- Have STABLE Resting Membrane Potentials
- Resting Membrane Potential (MP):
- Na+ & Ca+ channels are closed
- Any positive change to MP causes Fast Na+ channels to open → positive feedback → threshold
- Depolarisation:
- If MP reaches threshold, all Fast Na+ channels open;
- → Massive influx of Na+ into cell
- → Membrane depolarises
- Plateau:
- Fast Na+ channels inactivate
- → The small downward deflection is due to Efflux of K+ ions
- → Action potential causes membrane Voltage-Gated Ca+ channels to open
- This triggers further Ca+ release by the sarcoplasmic reticulum into the sarcoplasm (“Ca induced Ca Release”)
- This increased myoplasmic Ca+ causes muscular contraction.
- Plateau is sustained by influx of Ca+, balanced by efflux of K+ ions
- This triggers further Ca+ release by the sarcoplasmic reticulum into the sarcoplasm (“Ca induced Ca Release”)
- Repolarization:
- Influxing Ca+ channels close, the effluxing K+ channels remain open
- → Result is a net outward flow of positive charge. → Downward Deflection
- → As the MP falls, more K+ channels open, accelerating depolarization
- → Membrane Repolarizes & most of the K+ channels close
- Influxing Ca+ channels close, the effluxing K+ channels remain open
- What happens to the excess ions?
- Excess Na+ in the cell from depolarization is removed by the Na/K-ATPase
- Deficit of K+ in the cell from repolarization is replaced by the Na/K-ATPase
- Excess Ca+ from the Plateau Phase is eliminated by a Na/Ca Exchanger
- Resting Membrane Potential (MP):
NOTE:
There is considerable delay between myocardial contraction and the action potential.


Refractory Periods
In cardiac muscle, the Absolute Refractory Period continues until muscle relaxation
- Therefore, summation is not possible → tetany cannot occur (critical in heart)
- i.e., the depolarized cell will not respond to a 2nd stimulus until contraction is finished
Absolute Refractory Period
- Approximately 200ms
- Duration: from peak → plateau → halfway-polarized
Relative Refractory Period
- Na+ channels are closed – but can still respond to a stronger-than-normal stimulus
- Approximately 50ms
- Duration: last half of repolarization
The Sinoatrial (SA) Node
- The “Pacemaker” of the Heart: Unregulated Rate: 90-100bpm; however
- Parasympathetic NS lowers heart rate → keeps Normal Resting HR at 70bpm
- Sympathetic NS raises heart rate
- Location
- Posterior Wall of the Right Atrium near the opening of the Superior Vena Cava
- Nature of Action Potentials
- Continually Depolarizing 90-100bpm
- Takes 50ms for Action-Potential to reach the AV Node
- Role in Conduction Network
- Sets the pace for the heart as a whole
- Portion of Myocardium Served
- Contracts the Right & Left Atrium
The Atrioventricular (AV) Node
- 2nd in Command: Slower than the SA Node: 40-60bpm
- Location
- Inferior portion of the Interatrial Septum
- Directly above the Tricuspid Valve
- Nature of Action Potentials
- Continually Depolarizing – but slower than the SA Node (40-60 bpm)
- Role in Conduction Network
- To delay the impulse from the Sinoatrial Node → bundle branches;
- Delay allows the atria to empty their contents before Ventricular Contraction
- Delay: approx. 100ms
- Portion of Myocardium served
- Conducts the SA Node impulses to the Purkinje Fibers (which supply the ventricular walls)
Bundle Branches (Bundles of His)
- 3rd in Command: slower than AV & SA Nodes: 20–40 bpm
- Location
- Fork of branches – superior portion of interventricular septum
- Nature of Action Potentials
- Continually depolarizing – slower than AV & SA Nodes (20–40 bpm)
- Role in conduction network
- Serves as the only connection between the 2 atria & 2 ventricles
- The 2 atria & 2 ventricles are isolated by the fibrous skeleton and lack of gap junctions
- Portion of the myocardium served
- Transmits impulses from the AV node to the R&L Bundle branches
- Then along the interventricular septum → apex of the heart
Purkinje Fibers
- Specialized Myocytes with very few myofibrils → don’t contract during impulse transmission
- Location
- The Inner Ventricular Walls of the Heart – just below the Endocardium
- Begin at the heart apex, then turn superiorly into the Ventricular Walls
- Nature of Action Potentials
- Conductile
- Resembles those of Ventricular Myocardial Fibers
- However the Depolarisation is more pronounced & the Plateau is longer.
- Long Refractory period
- Capable of Spontaneous Depolarisation – 15bpm
- Role in Conduction Network
- Carry the contraction impulse from the L & R Bundle Branches to the Myocardium of the Ventricles
- Causes Ventricles to contract
- Portion of Myocardium served
- R & L Ventricles
| SA Node | AV Node | Bundles of His | Purkinje Fibers | |
| Description | “Pacemaker” of the Heart | 2nd in command | 3rd in command | Specialized myocytes with very few myofibrils (do not contract during impulse transmission) |
| Rate (bpm) | 90-100 | 40-60 | 20-40 | N/A |
| Location | Posterior wall of the right atrium; near opening of Superior Vena Cava | Inferior portion of the interatrial septum; directly above the tricuspid valve | Fork of branches; superior portion of interventricular septum | Inner ventricular walls of the heart (just below the endocardium); begin at the heart apex, then turn superiorly into the ventricular walls |
| Nature of Action Potentials | Continually depolarizing (90-100bpm); takes 50ms for action potential to reach AV node | Continually depolarizing (slower than the SA node) | Continually depolarizing (slower than AV & SA nodes) | Conductile (resembles ventricular myocardial fibers); more pronounced depolarization; longer plateau; longer refractory period |
| Role in Conduction Network | Sets the pace for the heart as a whole | Delay the impulse from the SA node → Bundle branches (100ms) | Serves as the only connection between the 2 atria and 2 ventricles (these are isolated by the fibrous skeleton and lack of gap junctions) | Carry the contraction impulse from the left and right Bundle branches to the myocardium of the ventricles; causes ventricles to contract |
| Portion of Myocardium Served | Contracts the right and left atria | Conducts the SA node impulses to the Purkinje Fibers | Transmits impulses from the AV node to the right and left Bundle branches; then along the interventricular septum → apex of the heart | Right and left ventricles |
Effect of the Autonomic Nervous System (ANS)
Although the heart can operate on its own, it normally communicates with the brain via the autonomic nervous system
Parasympathetic Nervous System
- Innervates SA & AV Nodes → slows heart rate
- Direct Stimulation → Releases Acetylcholine → Muscarinic receptors in SA/AV Nodes
- Causes increased K+ permeability (Efflux) → Hyperpolarizes the cell
- Cell takes longer to reach threshold → Lower Heart Rate
- Causes increased K+ permeability (Efflux) → Hyperpolarizes the cell
Sympathetic Nervous System
- Innervates the SA & AV Nodes & ventricular muscle
- → Raises Heart Rate
- → Increases force of contraction
- → Dilates arteries
- Indirect stimulation → Sympathetic nerve fibers release Noradrenaline (Norepinephrine) at their cardiac synapses → Binds to Beta 1 Receptors on Nodes & Muscles →
- Initiates a cyclic AMP pathway → Increases Na+ + Ca+ permeability in nodal tissue & increases Ca+ permeability(Membrane & SR) in muscle tissue
- Effects on Nodal Tissue
- ++Permeability to Na+ → more influx of Na+ → Membrane ‘drifts’ quicker to threshold → Increased heart rate
- ++Permeability to Ca+ → more influx of Ca+ → Membrane depolarisation is quicker → Increased heart rate
- Effects on Contractile Tissue
- ++ Membrane permeability to Ca+ → More influx of Ca+ →
- ++Sarcoplasmic reticulum permeability to Ca+ → Efflux of Ca+ into cytoplasm→
- Increases available Ca+ for contraction → Contractile force increases


Electrocardiogram (ECG) Physiology
What is an ECG?
- A recording of all action potentials by nodal & contractile cells in the heart at a given time
- It i NOT a single action potential
- A “lead” refers to a combination of electrodes that form an imaginary line in the body, along which the electrical signals are measured
- i.e., a 12 ‘lead’ ECG usually only uses 10 electrodes
- Measured by voltmeters → record electrical potential across 2 points
- 3x bipolar leads: measure voltages between the arms or between an arm and a leg
- I = LA(+) RA(-)
- II = LL(+) RA(-)
- III = LL(+) LA(-)
- 9x unipolar leads: look at the heart in a ‘3D’ image
- 3x bipolar leads: measure voltages between the arms or between an arm and a leg
- Graphic output
- X-axis: time
- Y-axis: amplitude (voltage) – proportional to number and size of cells
- Understanding waveforms
- When a Depolarisation Wavefront moves toward a positive electrode, a Positive deflection results in the corresponding lead.
- When a Depolarisation Wavefront moves away from a positive electrode, a Negative deflection results in the corresponding lead.
- When a Depolarisation Wavefront moves perpendicular to a positive electrode, it first creates a positive deflection, then a negative deflection.
How Each Wave Segment is Formed
| P-Wave Depolarization of the atria Presence of this wave indicates the SA node is working |
|
| PR-Segment Reflects the delay between SA & AV nodes Atrial contraction is occurring at this time |
|
| Q-Wave Interventricular septum depolarization Wave direction (see blue arrow) is perpendicular to the main electrical axis → results in a biphasic trace Only the negative deflection is seen due to signal cancellation by atrial repolarization Sometimes this wave isn’t seen at all |
|
| R-Wave Ventricular depolarization Wave direction (see blue arrow) is the same as the main electrical axis → positive deflection R-wave amplitude is large due to sheer numbers of depolarizing myocytes |
|
| S-Wave Depolarization of the myocytes at the last of the Purkinje Fibers Wave direction (black arrow) opposes the main electrical axis → negative deflection This wave is not always seen |
|
| ST-Segment Ventricular contraction is occurring at this time Due to the lag between excitation and contraction |
|
| T-Wave Ventricular repolarization Positive deflection despite being a repolarization wave – because repolarization waves travel in the opposite direction to depolarization waves |
Relating ECG Waves to Events in the Cardiac Cycle 
- Contractions of the heart ALWAYS lag behind impulses seen on the ECG
- Fluids move from high → low pressure
- Heart valves ensure a unidirectional flow of blood
- Coordinated contraction timing – critical for correct flow of blood
The Heart’s Electrical Axis
- Refers to the general direction of the heart’s depolarization wavefront (or ‘mean electrical vector’) in the frontal plane
- It is usually oriented in a ‘Right Shoulder to Left Leg’ direction
- Determining the electrical axis from an ECG trace
- 3 methods
- Quadrant Method (the one you’re concerned with)
- Peak Height Measurement Method
- The Degree Method
- 3 methods
The Quadrant Method
| LEAD 1 | LEAD aVF | QUADRANT | AXIS |
| + | + | Normal(0 to +90°) | |
| + | – | **Possible LAD (0 to -90°) | |
| – | + | RAD (+90° to 180°) | |
| – | – | Extreme Axis(-90° to 180°) |
- Normal Axis. QRS positive in I and aVF (0 90 degrees). Normal axis is actually 30 to 105 degrees.
- Left Axis Deviation (LAD). QRS positive in I and negative in aVF, 30 to 90 degrees
- Right Axis Deviation (RAD). QRS negative in I and positive in aVF, +105 to +180 degrees
- Extreme RAD. QRS negative in I and negative in aVF, +180 to +270 or 90 to 180 degrees
Algorithm for Looking at ECGs
- Check Pt ID
- Check voltage & timing
- 25mm/sec
- 1 large square = 0.2s (1/5sec)
- 1 small square = 0.04s
- What is the rate?
- 300/number of large squares between QRS Complexes
- Tachycardia >100bpm
- Bradycardia <60bpm
- 300/number of large squares between QRS Complexes
- What is the rhythm?
- Sinus? (are there P-Waves before each QRS complex)
- If not sinus?
- Is it regular?
- Irregular?
- Irregularly Irregular (AF)
- Brady/Tachy
- Atrial Fibrillation
- Irregularly Irregular
- P-Waves at 300/min
- QRS
- Is there one QRS for each P-wave?
- Long PR Interval? (1st degree heart block)
- Missed Beats? (Second degree block)
- No relationship? Complete heart block
- Look for QRS Complexes
- How wide – should be < 3 squares
- If wide – It is most likely Ventricular
- (Sometimes atrial with aberrant conduction (LBBB/RBBB)
- IF Tachycardia, & Wide Complex → VT is most likely. (If hypotensive → Shock; if Normotensive → IV Drugs)
- Look for T-Waves
- Upright or Inverted
- Look at ST-Segment
- Raised, depressed, or inverted
- ST Distribution → Tells you which of the coronaries are blocked/damaged
- Inferior ischaemia (II, III, AVF)
- Lateral ischaemia (I, II, AVL, V5, V6)
- Anterior ischaemia (V, leads 2-6)
- NOTE: Normal ECG Doesn’t exclude infarct.
- ST Depression → Ischaemia
- ST Elevation → Infarction
- If LBBB or Paced, you CANNOT comment on ST-Segment
Mechanical Events of the Cardiac Cycle
Structure-Function Relationship of the Heart
- The Myocardium is essentially one long muscle orientated in a spiral-like fashion
- This allows the heart to be electrically integrated
- Allows the heart to ‘wring out’ the blood within it
- This setup facilitates a strong pumping action
Terms
- Systole = Myocardial Contraction
- Diastole = Myocardial Relaxation
- Stroke Volume = Output of Blood from the heart per contraction (≈80mL of blood)
- Heart Rate = #Heart Beats/Minute
- Cardiac Output
- Volume of blood ejected from the heart per minute (Typically ≈5L/min)
- Cardiac Output = Heart Rate x Stroke Volume
- Chronotropic Influences
- Affect heart rate
- Inotropic Influences
- Affect contractility (& stroke volume)
- Dromotropic Influences
- Affect AV Node delay
- End Diastolic Volume = Ventricular volume at end of Diastole (When ventricle is fullest)
- End Systolic Volume = Ventricular volume after Contraction (Normal ≈ 60-65%)
- Preload = The degree of stretching of the heart muscle during Ventricular Diastole
- (↑Preload = ↑cross linking of myofibrils = ↑Contraction (“Frank Starling Mechanism”)
- Afterload = The ventricular pressure required to eject blood into aorta/pulmonary artery
- (↑Afterload = ↓SV due to ↓ejection time)
Overview of the Cardiac Cycle
Phase 1 – Atrial Contraction (Systole) + Ventricular Filling (Diastole)
- Contraction of atria
- → intra-atrial pressure increases
- → blood pushed into ventricles through AV valves
- Note: ventricles are already 70% full from passive venous filling
- At the end of atrial systole, ventricles have EDV (end diastolic volume) ≈130mL
Phase 2 – Ventricular Systole
AV Valves Close
- Ventricular pressure exceeds atrial pressure → AV valves shut
- Brief period of isovolumetric contraction
- Where ventricular pressure rises, but volume stays constant
- The beginning of ventricular systole
- All valves are still closed
Semilunar Valves Open
- Ventricular pressure exceeds aortic/pulmonary pressure → blood ejected
- ≈80mL of blood ejected each time (Stroke Volume)
- Ventricular volume decreases
Semilunar Valves Close
- Ventricular pressure then falls below aortic/pulmonary pressure → semilunar valves close
- Sudden closure of semilunar valves causes the dicrotic notch
- Result of elasticity of the aorta & blood rebounding off the closed SL valve
- Causes a slight peak in aortic pressure
- Sudden closure of semilunar valves causes the dicrotic notch
- Note: ventricles never fully empty
- ESV (End Systolic Volume) = amount of blood left in ventricles → 50mL
Phase 3 – Ventricular Diastole
- Ventricles relax → Ventricular pressure falls below atrial pressure → AV valves open:
- Blood → from Atria into ventricles
- (NOTE: Passive filling from venous return is responsible for 70% of ventricular filling.)
Cardio-Dynamics
Cardiac Output
- Useful when examining cardiac function over time
- Determined by 2 things
- Stroke Volume
- Heart Rate
Cardiac Output(mL/min) = Stroke Volume X Heart Rate
- Average CO ≈ 5L/min (i.e.: The entire blood supply circulates once per minute)
- Cardiac Output is regulated such that peripheral tissues receive adequate blood supply
Heart Rate
- Depends on tissue-satisfaction with nutrients and O2
- Terms
- Bradycardia. HR slower than normal (too fast → stroke volume & CO suffer)
- Tachycardia. HR faster than normal
5 Things that Affect Heart Rate
- Alterations in SA Node Firing
- SA node is the pacemaker; therefore, change it rate → change heart rate
- → change CO
- SA node is the pacemaker; therefore, change it rate → change heart rate
- Autonomic Nervous System
- Parasympathetic (Vagus Nerve)
- Decrease HR (negative chronotropic effect)
- Increase AV node delay (negative dromotropic effect)
- NOTE: ONLY A TINY EFFECT ON CONTRACTILITY
- Sympathetic (Sympathetic Chains)
- Increase HR (positive chronotropic effect)
- Increase force of contraction (positive inotropic effect)
- Parasympathetic (Vagus Nerve)
- Reflex Controls
- Bainbridge Reflex (Atrial Walls)
- Where an ↑Venous Return → ↑Heart Rate
- (Stretch of Atrial Walls → Stretch Receptors → Sympathetic NS → ↑HR)
- Responsible for 40-60% of HR increases
- Chemoreceptor Reflex
- ↓Low O2 or ↑CO2 in Peripheral-Tissue → ↑HR & ↑Respiratory Rate
- Baroreceptor Reflex (Aortic & Carotids)
- Where an ↑BP → ↓HR & ↓Contractility (+ Vasodilation)
- 2 Main Baroreceptors
- Aortic → Vagus Nerve → CV Center (medulla/pons)
- Carotid → Hering’s Nerve → CV Center (medulla/pons)
- Constantly responds to blood pressure change
- (via stretch in vessel walls)
- More stretch = More firing: leads to:
- Parasympathetic activation
- Sympathetic deactivation
- Receptors never silent – constantly signaling
- Quick to respond
- In hypertension → receptors recalibrate to the higher BP
- Changes HR accordingly
- Bainbridge Reflex (Atrial Walls)
- Atrial Node Stretching (similar to baroreceptor reflex, but in the atrium)
- Venous return fills atria with blood
- When Venous Return ↑, Atrial Walls Stretch → Stretches SA-Node
- Stretching of SA node cells → More rapid depolarisation → ↑HR
- Responsible for 15% of HR increases
- Influenced by
- Arterial Pressure
- Peripheral Compliance
- Local Blood Flow
- Capillary Exchange
- Venous return fills atria with blood
- Chemical Regulation
- Hormones
- Adrenaline
- Thyroxine
- Insulin
- Ions
- Na+
- K+
- Ca2+
- Hormones
Other Factors that Affect HR
- Age (old → lower resting HR)
- Gender (females → higher resting HR)
- Physical fitness (fit → lower resting HR)
- Temperature (hot → higher resting HR)
Stroke Volume
- Blood output per heartbeat
- Useful when examining the efficiency of a single cardiac cycle
Stroke Volume (SV) = End Diastolic Volume (EDV) – End Systolic Volume (ESV)
- Therefore, Stroke Volume is ↑ by
- ↑ Ventricular filling time (duration of ventricular diastole)
- ↑ Venous return
- ↓ Arterial BP (a high arterial BP → harder to eject blood → ESV increases)
- ↑ Force of ventricular contraction
2 Things that Affect Stroke Volume
Preload
- The degree of stretching of the heart muscle during Ventricular Diastole
- Caused by amounts of blood from venous return
- Influenced by
- Arterial Pressure
- Peripheral Compliance
- Local Blood Flow (depending on the demands of those tissues)
- Capillary Exchange.
- Preload ↑ as EDV↑ (directly proportional)
- ↑End Diastolic Volume = ↑Stroke Volume (Frank-Starling Law)
- Affects % of actin/myosin contact in myocytes→ Affects cross-bridge cycling:
- → Affects muscle’s ability to produce tension
- Preload varies with demands placed on the heart
- Contractility
- Inotropy
- Force produced during contraction at a given preload
- Influences End Systolic Volume (↑Contractility = ↓ESV)
Afterload
- Back pressure exerted by arterial blood
- The tension needed by ventricular contraction to open semilunar valve
- i.e., The pressure the heart must reach to eject blood
- ↑Afterload = ↑ESV = ↓SV
- Afterload is increased by anything that restricts arterial blood flow
Hemodynamics
Relationship between Flow, Pressure, Resistance
- Flow is directly proportional to pressure gradient between 2 points (change in pressure)
- Flow is inversely proportional to resistance
- Resistance is far more important in determining local blood flow versus the pressure gradient
Blood Flow Rate
- The Amount of blood flowing through a vessel/organ/system per unit time (mLs/min)
- Determined by pressure gradient & resistance, NOT velocity
- Systemic Blood Flow = Cardiac Output (relatively constant)
- Specific Organ Blood Flow – may vary widely due to its immediate needs
Velocity of Flow
- Velocity of Flow = SPEED of flowing blood (mm/sec)
- e..g, A constricted vessel will have a lower flow rate, but a higher velocity of flow (i.e., Garden hose)
- Note: Velocity tends to change by a greater magnitude than the change in Flow Rate
Blood Pressure
- The Pressure exerted on the vessel wall by contained blood (mmHg)
- Decreases with distance from heart (arterial system)
- Decreases with 10%+ decrease blood volume
- Increases with vessel constriction (provided same blood volume)
Resistance
- The amount of friction blood encounters as it passes through the vessels
- 3 factors influencing resistance
- Blood Viscosity (↑Viscosity = ↑Resistance) (Fairly Constant)
- Total Vessel Length (longer vessel = ↑ resistance) (Fairly Constant)
- Vessel Diameter (thinner vessel = ↑resistance) (Frequently Changes)
- Most responsible for changes in BP
- Systemic Vascular Resistance = Combination of the Above Factors
Effects of Vessel Diameter (Vasomotion) on Flow Rate
- The flow rate is directly proportional to the 4th power of the vessel diameter
- i.e., Small changes in vessel diameter → Changes flow rate by an exponent of 4
- Poieuille’s Law
Effects of Vessel Diameter (Vasomotion) on Flow Velocity
- Flow rate is inversely proportional to the vessel’s cross-sectional area
- i.e., An ɑ x increase in cross-sectional area → decrease in flow velocity by a factor of ɑ
Blood Pressure Physiology
Factors Influencing Blood Pressure
- Cardiac Output
- ↑Cardiac Output = ↑ BP
- Peripheral Resistance
- Causes back pressure in blood (arterial system)
- e.g., In obesity, peripheral resistance increases.
- Blood Volume
- (assuming constant vessel diameters) ↑Blood Volume = ↑BP
- Its effect depends on vessel compliance
BP = Cardiac Output X Total Peripheral Resistance
Types of Blood Pressure
Systolic
- Peak aortic pressure reached during ventricular systole
- A function of
- Peak rate of ejection
- Vessel wall compliance
- Diastolic BP
- Normal = 120mmHg
Diastolic
- Lowest aortic pressure reached during ventricular diastole, due to blood left after peripheral runoff
- A function of
- Blood volume
- Heart rate
- Peripheral resistance
- Normal = 80mmHg
Pulse Pressure*
- Pulse Pressure = Systolic Pressure – Diastolic Pressure
- e.g., 120mmHg – 80mmHg
- Normal = 40mmHg
- If lower: may be an indication of Aortic Stenosis or Atherosclerosis (slowed peripheral runoff)
Mean Arterial Pressure*
- MAP = Diastolic Pressure + 1/3(Pulse Pressure)
- The pressure that propels blood to the tissues – maintains tissue perfusion
- Maintains flow through capillary beds
- Must be high enough to overcome peripheral resistance (if not, blood doesn’t move)
- Finely controlled
3 Main Regulators of Mean Arterial Pressure
Autoregulation (at the tissue level)
- Localized automatic vasodilation/constriction at the tissue level
- Allows control of flow within a single capillary bed
- Ensures perfusion of the ‘needy’ tissues
- Metabolic controls → Vasodilation
- Low oxygen/nutrient levels
- Nitric Oxide
- Endothelin
- Inflammatory chemicals: histamine/kinins/prostaglandins
- Myogenic control → Vasoconstriction
- Sheer stress: Vascular smooth muscle responds to passive stretch (↑vascular pressure) with increased tone
- Prevents excessively high tissue perfusion that could rupture smaller blood vessels
- Reduced stretch promotes vasodilation → flow increases
- Sheer stress: Vascular smooth muscle responds to passive stretch (↑vascular pressure) with increased tone
Neural Mechanisms
- Vasomotor Center (medulla)
- Take info from receptors
- Baroreceptors (primarily)
- Chemoreceptors (lesser degree)
- Transmit impulses via sympathetic nervous system
- ↑ sympathetic activity = vasoconstriction = ↑ BP
- ↓ sympathetic activity = vasodilation = ↓ BP
- Take info from receptors
- Cardiovascular Centers of the ANS
- Sympathetic → ↑HR & Contractility → ↑MAP
- Parasympathetic → ↓Heart Rate → ↓MAP
Endocrine Mechanisms (Kidney Level)
- More for long-term BP & blood-volume regulation
- Antidiuretic Hormone (ADH)
- Aka vasopressin
- Released due to low blood volume
- ADH → Water Retention Increased → ↑MAP
- Angiotensin II
- Released due to low blood pressure
- Potent vasoconstrictor
- Increases cardiac output & blood volume
- Angiotensin II → Vasoconstriction → ↑MAP
- NOTE: ‘ACE’ (Angiotensin I Converting Enzyme) activates it to Angiotensin II. Hence ‘ACE-Inhibitors’ are often used as AntiHypertension medicine)
- Erythropoietin
- Released due to low pressure & low O2 levels
- Increases RBC production to increase blood volume
- EPO → Hematopoiesis → ↑Blood Volume → ↑MAP
- Natriuretic Peptides (Released by the heart)
- Released by the heart due to high blood pressure & volume
- ↑Stretch on Heart → NP Release → ↑Diuresis → Reduces BP & Volume
- Also inhibits ADH & Angiotensin II → Reduces BP & Volume
Anatomy & Physiology of Blood Vessels
Introduction to Blood Vessels
3 Classes
- Arteries → carry blood away from the heart
- Elastic Arteries
- e.g., Aorta & major branches (Conducting Vessels)
- Muscular Arteries
- e.g., Coeliac trunk & renal arteries (Distributing Vessels)
- Arterioles
- e.g., Intra-organ arteries (Resistance Vessels)
- Terminal Arteriole
- e.g., Afferent arteriole in kidney
- Elastic Arteries
- Capillaries → intimate contact with tissue; facilitate cell nutrient/waste transfer
- Vascular shunt
- True capillaries
- Veins → carry blood back to the heart
- Post-capillary venule (union of capillaries)
- Small veins & large veins
- Capacitance vessels
- 65% of body’s blood is venous
Relationships between Vessel Diameter, Cross-Sectional Area, Local Blood Pressure, and Velocity of Flow
Blood Vessel Structure
3-layered wall
Tunica Intima
- i.e., The layer in intimate contact with the blood (luminal)
- Consists of the endothelium (simple squamous epithelium)
- Larger vessels also have a sub-endothelial layer
Tunica Media
- Middle and thickest layer (smooth muscle + elastin)
- Circulating smooth muscle
- Sheets of elastin
- Regulated by sympathetic nervous system + chemicals
- Contraction/dilation maintains blood pressure
Tunica Externa
- Outermost layer (Loose collagen fibers)
- NOTE: Also contains nerve fibers, lymphatics, and vasa vasorum (in larger vessels)
The Arterial System
Elastic (Conducting) Arteries
- The aorta + its major branches
- Thick-walled
- Large lumen = low resistance
- Highest proportion of elastin
- Withstands pressure fluxes
- Smooths out pressure fluxes
- ‘Stretch’ = potential energy → helps propel blood during diastole
Muscular (Distributing) Arteries
- Distal to elastic arteries
- Deliver blood to specific body organs
- Diameter: 0.3mm→1cm
- Thickest tunica media
- Due to smooth muscle
- Highest proportion of smooth muscle
- Are active in vasoconstriction
- Are therefore less distensible (less elastin)
Arterioles
- Smallest arteries
- Larger arterioles have all 3 tunics (intima/media/externa)
- Most of the tunica media is smooth muscle
- Smaller Arterioles
- Lead to capillary beds
- Little more than 1 layer of smooth muscle around the endothelial lining
- Autoregulation of diameter
- Controlled by
- Neural (electrical) signals
- Hormonal signals
- Noradrenaline
- Epinephrine
- Vasopressin
- Endothelin-1
- Local chemicals
- Controls blood flow to capillary beds
- When constricted – tissues served are bypassed
- When dilated – tissues served receive blood
- Controlled by
- Biggest controller of blood pressure
The Capillary System
- Smallest blood vessels – microscopic
- Thin, thin walls
- Tunica intima only (i.e., only 1 layer thick)
- Average length = 1mm
- Diameter: the width of a single RBC
- RBC’s flow through capillaries in single file
- RBC’s shape allows them to stack up efficiently against each other
- Penetrate most tissues, except:
- Tendons
- Ligaments
- Cartilage
- Epithelia
- Main role
- Exchange of gases/nutrients/hormones/wastes
- Exchange occurs between blood & interstitial fluid
Capillary Beds
- Capillaries are only effective in large numbers
- Form networks called ‘capillary beds’
- Facilitates microcirculation
- Blood flow from an Arteriole → Venule
- Consist of 2 types of vessels
- Vascular Shunt
- From metarteriole → thoroughfare channel
- Short vessel – directly connects arteriole with venule
- True Capillaries
- The ones that actually take part in exchange with tissues
- Usually branch off the metarteriole (proximal end of vascular shunt)
- Return to the thoroughfare channel (distal end of vascular shunt)
- Precapillary Sphincters
- Smooth muscle cuffs
- Surround the roots of each true capillary (arterial ends)
- Regulates blood flow into each capillary
- i.e., Blood can either go through capillary or through the shunt
- Vascular Shunt
- A capillary bed may be flooded with blood or bypassed, depending on conditions in that organ
3 Types of Capillaries
Continuous Capillaries
- ‘Continuous’ = uninterrupted endothelial lining
- Adjacent cells form intercellular clefts
- Joined by incomplete-tight-junctions
- i.e., Allow limited passage of fluids & solutes
- NOTE: In the brain, the tight-junctions are complete → blood brain barrier
Fenestrated Capillaries
- Endothelial cells are riddled with oval pores (fenestrations = windows)
- Much more permeable to fluids & solutes than continuous capillaries
- Abundant wherever active absorption/filtration occurs
- Intestines
- Kidneys
- Endocrine organs (allow hormones rapid entry to blood)
Sinusoids (Sinusoidal Capillaries)
- aka “leaky capillaries”
- Found ONLY in
- Liver
- Bone marrow
- Lymphoid tissues
- Some endocrine organs
- Large irregularly-shaped lumens
- Usually fenestrated
- ‘Discontinuous’ = interrupted by Kupffer cells
- Remove & destroy bacteria
- Intercellular clefts → larger + have fewer tight junctions
- Allow large molecules & leukocytes passage through to interstitial space
The Venous System
- Vessels carry blood back towards the heart (from capillary beds)
- Vessels gradually increase in diameter & thickness towards the heart
2 Types
Venules
- Formed by union of capillaries (post-capillary venules)
- Consist entirely of endothelium
- Extremely porous
- Allows passage of
- Fluid
- White blood cells (migrate through wall into inflamed tissue)
- The larger venules
- Have 1 or 2 layers of smooth muscle (i.e., tunica media)
- Have a thin tunica externa as well
Veins
- Formed by union of venules
- 3 distinct tunics (but walls thinner than corresponding arteries)
- Thinner walls due to lower blood pressure
- Tunica media
- Poorly developed
- Some smooth muscle
- Some elastin
- Tend to be thin even in large veins
- Tunica externa
- Heaviest layer (thicker than media)
- Thick longitudinal collagen bundles
- Thick elastic networks
- Lumens larger than corresponding arteries
- The reason 65% of the body’s blood is in the veins
- Therefore veins: aka “capacitance vessels”
- Lower blood pressure than arteries
- Require structural adaptations to get blood → heart:
- Large lumen (low resistance)
- Valves
- Require structural adaptations to get blood → heart:
- Venous valves
- Folds of tunica intima (resemble semilunar valves)
- Prevent blood flowing backward
- Ensures unidirectional flow
- Often have to work against gravity
- If faulty, causes thrombosis (e.g., varicose veins)

Fetal Circulation
- “Bypasses” / “shunts” of fetal circulatory system
- All of these “shunts” are occluded at birth due to pressure changes
Ductus Venosus
- Directs the oxygenated blood from the placental vein into inferior vena cava → heart
- Partially bypasses the liver sinusoids
Foramen Ovale
- An opening in the interatrial septum loosely closed by a flap of tissue
- Directs some blood entering the right atrium into the left atrium → aorta
- Partially bypasses the lungs
- NOTE: Foramen ovale can take up to 6 months to close
Ductus Arteriosus
- Directs most blood from right atrium of the heart directly into aorta
- Partially bypasses the lungs
Fluid Movements across a Vessel
- Determined by the balance of 2 forces
- Hence, fluid is forced out at arterial end, and reabsorbed at venous end
- The amount of fluid forced out = determined by the balance of net hydrostatic & osmotic forces
- i.e., Net Filtration Pressure = Net Hydrostatic Pressure – Net Osmotic Pressure
Capillary Hydrostatic Pressure
- The force the blood exerts against the capillary wall
- Hydrostatic pressure = capillary blood pressure ≈ 35 mmHgArterial End /15 mmHgVenous End
- Tends to force fluids through the capillary’s intercellular clefts (between endothelial cells)
- Capillary hydrostatic pressure drops as blood flows from arteriole → venule
- Net hydrostatic pressure = Capillary Hydrostatic Pressure – Interstitial Hydrostatic Pressure
- NOTE: Interstitial Hydrostatic Pressure ≈ 0 mmHg
Colloid Osmotic Pressure
- Opposes hydrostatic pressure
- Due to large, non-diffusible molecules (plasma proteins) drawing fluid into capillaries
- Typically ≈ 25 mmHg
- Relatively constant at both arterial & venous ends
- Net Osmotic Pressure = Capillary Osmotic Pressure – Interstitial Osmotic Pressure
- NOTE: Interstitial Osmotic Pressure ≈ 1 mmHg
Edema
- Abnormal accumulation of fluid in the interstitial space (i.e., tissue swelling)
- Caused by increase in flow of fluid → out of vessel or lack of reabsorption → into blood vessel
- Usually reflects an imbalance in colloid osmotic pressure on the 2 sides of the capillary membrane
- e.g., Low levels of plasma protein (reduces amount of water drawn into capillaries
- Contributing factors
- High BP (Hydrostatic Pressure)
- Can be due to incompetent valves
- Localized blood vessel blockage
- Congestive heart failure (pulmonary edema – due to blockage in pulmonary circuit)
- High blood volume
- Capillary Permeability
- Usually due to an inflammatory response
- High BP (Hydrostatic Pressure)
Injuries to Blood Vessels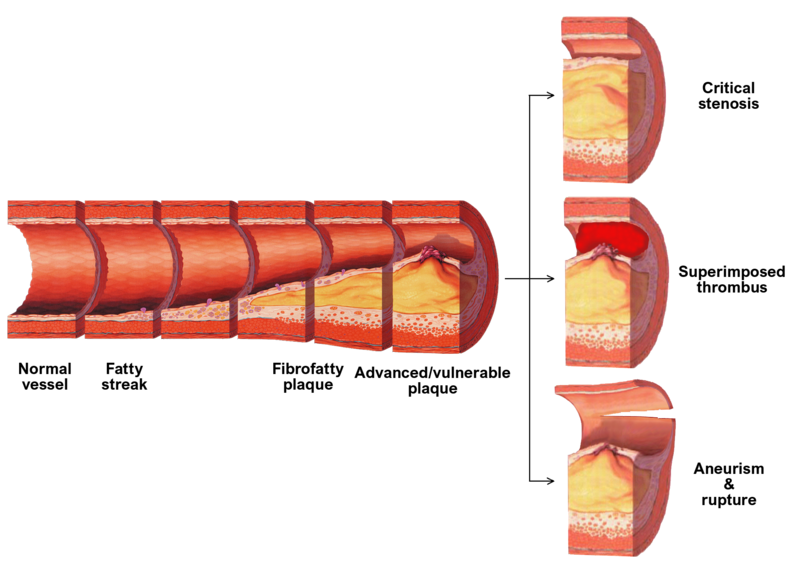
Atherosclerosis
- Formation of fatty plaques in the subendothelial layer
- Fatty plaques begin to ulcerate
Aneurysms
- Elastic arteries can lose their elasticity
- Due to having thinner walls, they’re more prone to aneurysm formation (bulging & potentially rupturing)
- Result in pooling of blood → eventual rupture
Dissections
- Blood builds up between the layers of the wall & eventually press the vessel closed
Physiology of Hypertension
What is Hypertension?
- Consistent diastolic of +90mmHg AND/OR
- Consistent systolic of +140mmHg
- A risk factor for other diseases
- Coronary artery disease
- Stroke
- Heart failure
- Renal failure
- Peripheral vascular disease
- Usually asymptomatic
- Often misdiagnosed due to
| Factor | Effect on BP Reading |
| Cuff – too wide/long | Lower than actual |
| Cuff – too narrow/short | Greater than normal |
| Arm – above heart | Lower than normal |
| Arm – below heart | Greater than normal |
| Arm – unsupported | Greater than normal |
| Respiration rate | Lower during inspiration |
| “White coat” phenomenon | Much greater than normal |
| Smoking/caffeine/activity 30 minutes prior to reading | Greater than normal |
Classifications Based on BP Ranges
| Category | Systolic | Diastolic | % Population |
| Normal | <130 | <85 | 83 |
| Pre-hypertensive | 130-139 | 85-89 | |
| Stage 1 Hypertension | 140-159 | 90-99 | 13.5 |
| Stage 2 Hypertension | 160-179 | 100-109 | 2 |
| Stage 3 Hypertension | 180-209 | 110-119 | |
| Stage 4 Hypertension | >210 | >120 | 1 |
2 Types of Hypertension (based on etiology)
Primary (Essential) Hypertension
- 90-95% of cases
- No specific cause
- Related to
- Obesity
- ↑Cholesterol
- Atherosclerosis
- ↑Salt Diet
- Diabetes
- Stress
- Family History
- Smoking
- Diastolic Hypertension
- Elevated diastolic pressure
- Relatively normal systolic (or slightly elevated)
- Mostly middle-aged men
- Isolated Systolic Hypertension
- Elevated systolic pressure
- Normal diastolic pressure
- i.e., High Pulse Pressure
- In older adults (60 years+)
- May be due to reduced compliance of the aorta with increasing age
- In younger adults (17-25 years)
- May be due to overactive sympathetic NS → ↑Cardiac Output
- Or congenitally stiff/narrow aorta
Secondary (Inessential) Hypertension
- 5-10% of cases
- Secondary to another disease, e.g.:
- Renal disease
- Endocrine disorders
- Pregnancy (pre-eclampsia)
- In 10% of pregnancies
- 20 weeks of gestation
- Others: cancer, drugs, alcohol
Organ Damage Caused by Hypertension
Heart
- Increased afterload
- ↑ Workload of Heart → ↑Afterload → Pumps Harder → Hypertrophy → Failure
- Left ventricular hypertrophy
- To compensate for higher workload
- Compromised L-Ventricular Volume → ↓Stroke Volume →↓Cardiac Output
Lungs
- Pulmonary congestion
- Backing up of blood in pulmonary circuit
- Why? ↑BP = ↑Aortic-BP = ↑Afterload = ↓SV = ↑ESV = ↓Pulmonary Blood Flow
Cerebrovascular
- Stroke – typically intracerebral hemorrhage
- Rupture of artery/arterioles in the brain
See image on right-hand side
Aorta/Peripheral Vascular
- Arterial mechanical damage (e.g., aneurysms/dissecting aneurysms)
- Accelerated atherosclerosis
Kidneys
- Nephrosclerosis (hardening of kidney blood vessels)
- Renal failure
Short-term Physiological Control of Blood Pressure
The Baroreceptor Reflex
Risk Factors of Hypertension
Age
- Blood pressure normally increases with age
- Baby: 50/40
- Child: 100/60
- Adult: 120/80
- Aged: 150/85 (quite normal)
- Due to the loss of elasticity of blood vessels (decreased compliance)
- Atherosclerosis
Race
Obesity
- Fatty diet → atherosclerosis
- Body fat → kms more vessels → ↑Peripheral Resistance → Hypertension
- Physical weight of fat may impede venous return
- Kidney dysfunction → loss of long-term BP (blood volume) control
Excess Na+ Intake
- If normal kidney function
- Na+ intake → slight BP increase (due to fluid retention)
- But excess Na+ intake and H2O excreted by kidneys → BP returns to normal
- If impaired kidney function
- Na+ intake → larger BP increase
- Because excess Na+ and H2O not excreted by kidneys (less efficiently)
Basic Hypertension Treatment Plan
Anti-Hypertensive Drug Mechanisms
- Diuretics
- Increase urination → decrease blood volume
- Aim: reduce workload on heart by reducing preload
- Sympatholytics
- Reduces sympathetic activity (Prevents ↑HR/↑Contractility = Decrease in CO)
- e.g., beta blockers
- Vasodilators
- Reduce peripheral resistance
- Reduce afterload
- Reduce workload on the heart
- Renin-Angiotensin Antagonists (ACE Inhibitors)
- Decreases effects of Renin-Angiotensin System
- Decreases sympathetic drive
- Decreases vasoconstriction
- Decreases fluid retention
- Decreases preload
- Decreases afterload
- Decreases effects of Renin-Angiotensin System
Physiology of Shock
Shock
- Profound hemodynamic/metabolic disorder due to inadequate blood flow and O2 delivery
- Common causes of shock
- Hypovolemic change
- Severe dehydration
- Hemorrhage
- Cardiogenic change
- Heart failure (heart isn’t getting enough blood out)
- Decreased venous return
- Distributive alteration
- Excessive metabolism (i.e., even a normal CO is inadequate)
- Abnormal perfusion patterns (i.e., most of CO perfuses tissues other than those in need)
- Neurogenic shock (i.e., sudden loss of vasomotor tone → massive venodilation)
- Anaphylactic shock (drastic decrease in CO & BP due to allergic reaction)
- Septic shock (disseminated bacterial infection in the body → extensive tissue damage)
- Hypovolemic change
3 Stages of Shock
Non-Progressive
- Stable, not self-perpetuating
- Symptoms
- Hypotension (Low BP)
- Tachycardia (High HR – body’s attempt to compensate for poor perfusion)
- Tachypnea (High breathing-rate – phrenic nerve stimulation – diaphragm)
- Oliguria (Low urine production by kidney)
- Clammy skin
- Chills
- Restlessness
- Altered consciousness
- Allergy symptoms (if anaphylaxis)
- The body’s compensatory mechanisms (below) will prevail without intervention.
- Aim to increase BP
Progressive Stage
- Unstable, vicious cycle of cardiovascular deterioration – self-perpetuating
- Compensatory mechanisms are insufficient to raise BP
- Perfusion continues to fall → organs become more ischemic (heart → failure)
- Cardiac depression (due to O2 Deficit to Heart)
- Vasomotor failure (due to O2 Deficit to Brain)
- “Sludged blood” (viscosity ↑ – harder to move)
- Increased capillary permeability
- Symptoms
- Beginning of organ failure
- Severely altered consciousness
- Marked bradycardia (initially tachycardic – but now the body is giving up)
- Tachypnea (fast breathing) with dyspnea (no breathing)
- Cold, lifeless skin
- Acidosis – (CO2 equation affected)
- Treatment
- Identify & remove causative agents
- Volume replacement for hypovolemia
- If septic shock: antibiotics
- Sympathomimetic drugs: if neurogenic shock (loss of vasomotor tone – vasodilation)
- Fatal if untreated
